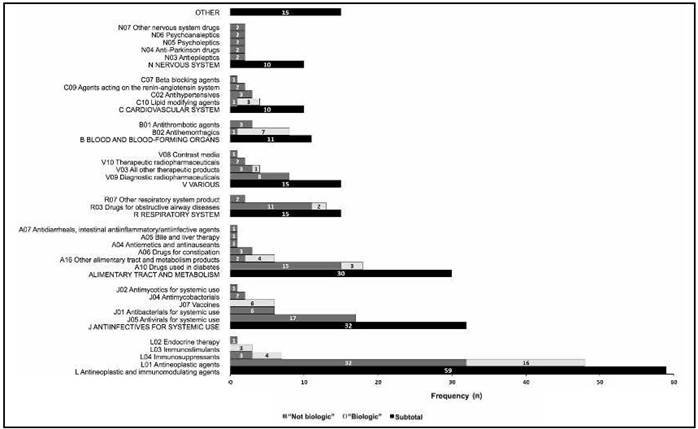
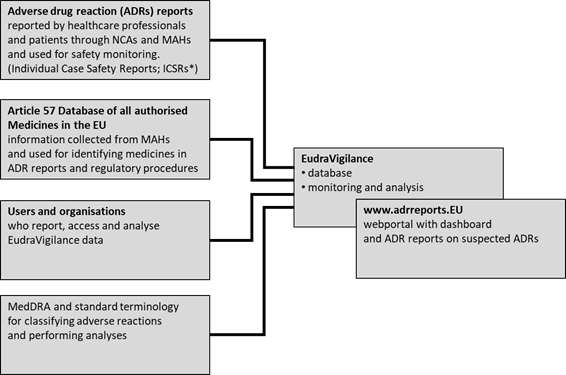
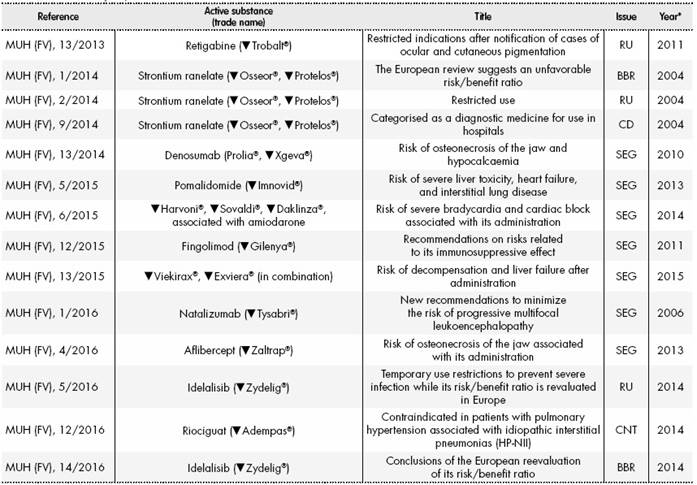
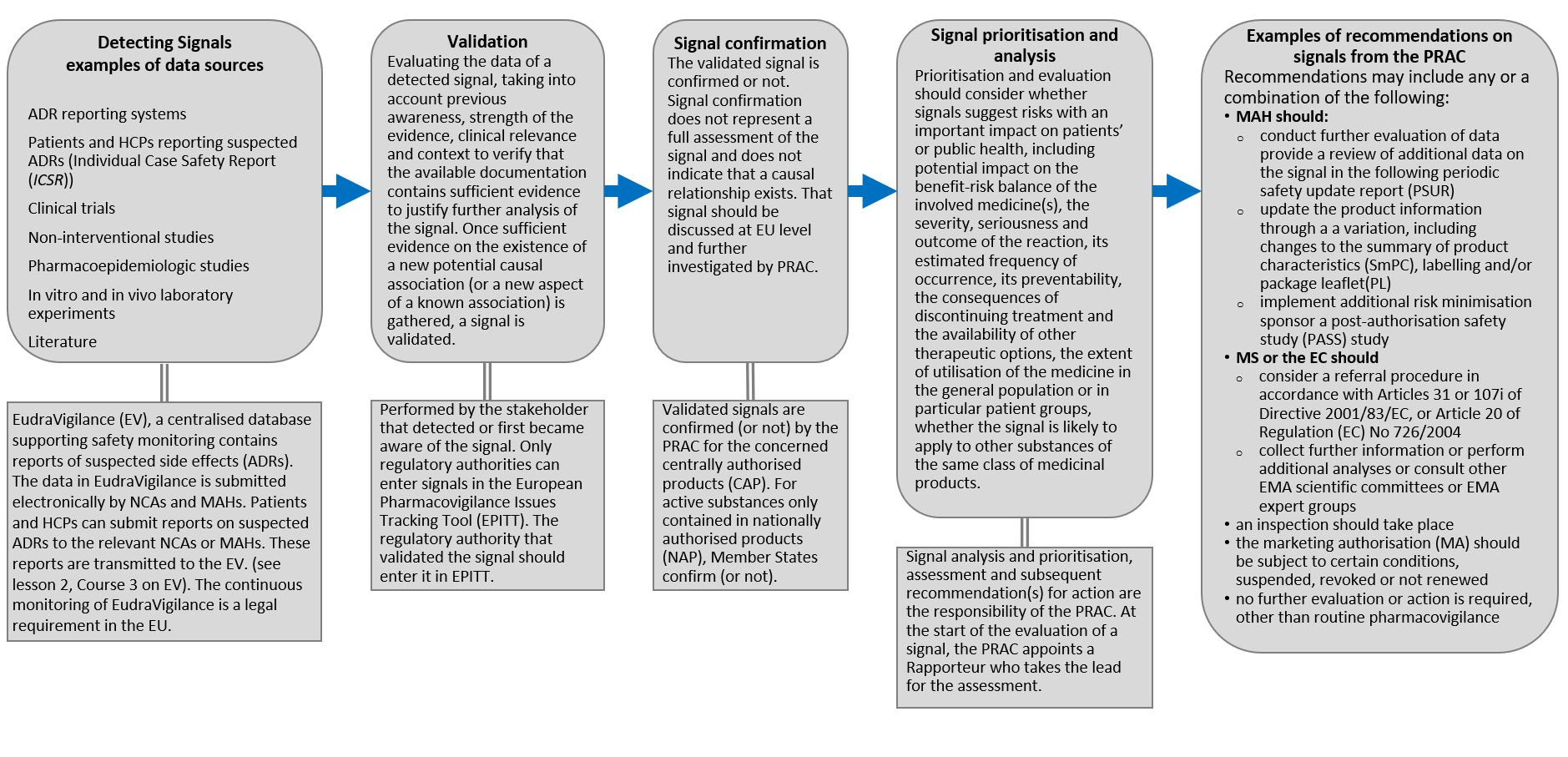
Improving the Safety of Medicines in the European Union: From Signals to Action - Potts - 2020 - Clinical Pharmacology & Therapeutics - Wiley Online Library

EU Commission Report on National and EMA experience on Medicines subject to Additional Monitoring released today

EMA updates SmPC and package leaflet templates with new black symbol | Signs & Symptoms of Translation
IRISH MEDICINES BOARD ADDITIONAL MONITORING: BLACK TRIANGLE INTRODUCED AS PROMPT TO REPORT SUSPECTED ADVERSE REACTIONS

Monitoring evidence on overall survival benefits of anticancer drugs approved by the European Medicines Agency between 2009 and 2015 - ScienceDirect