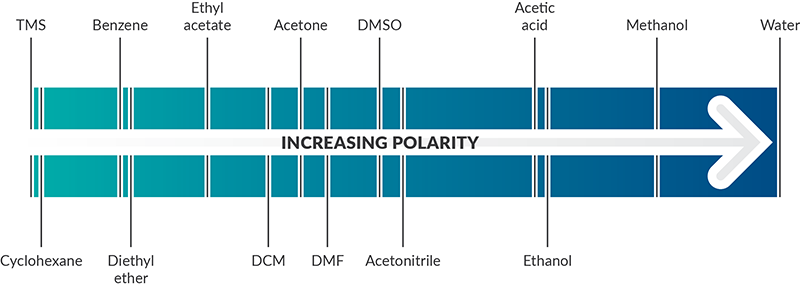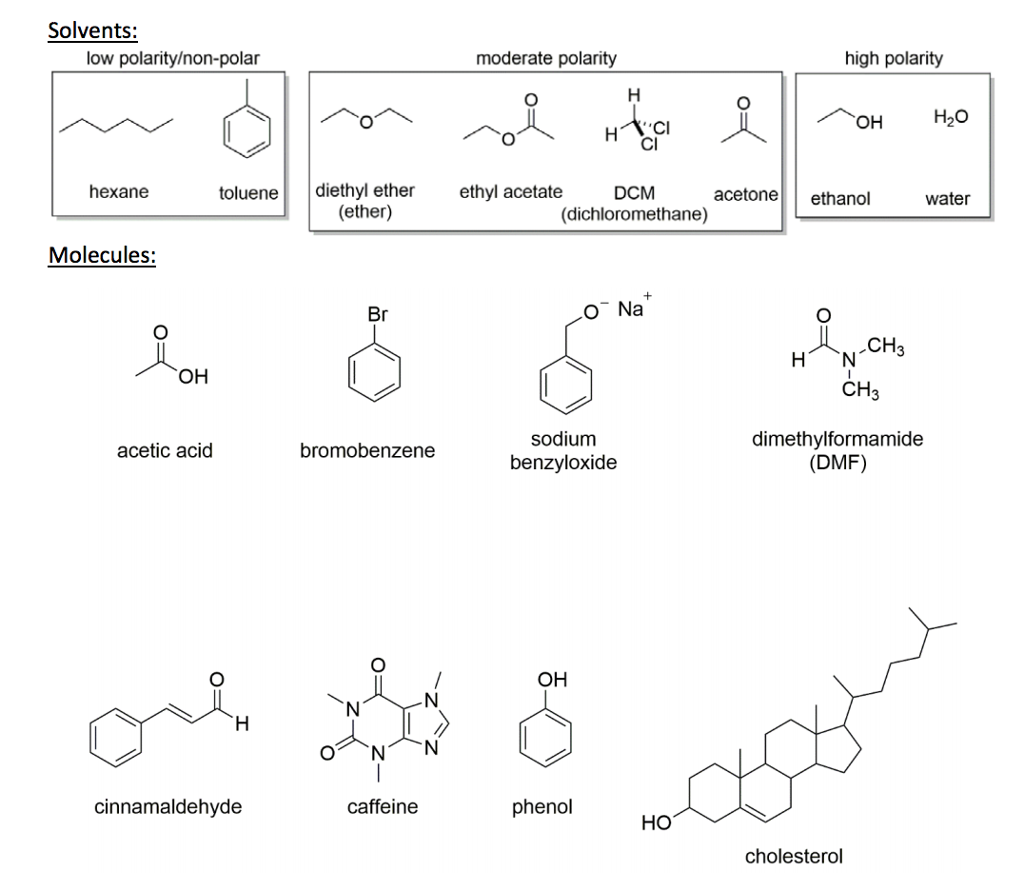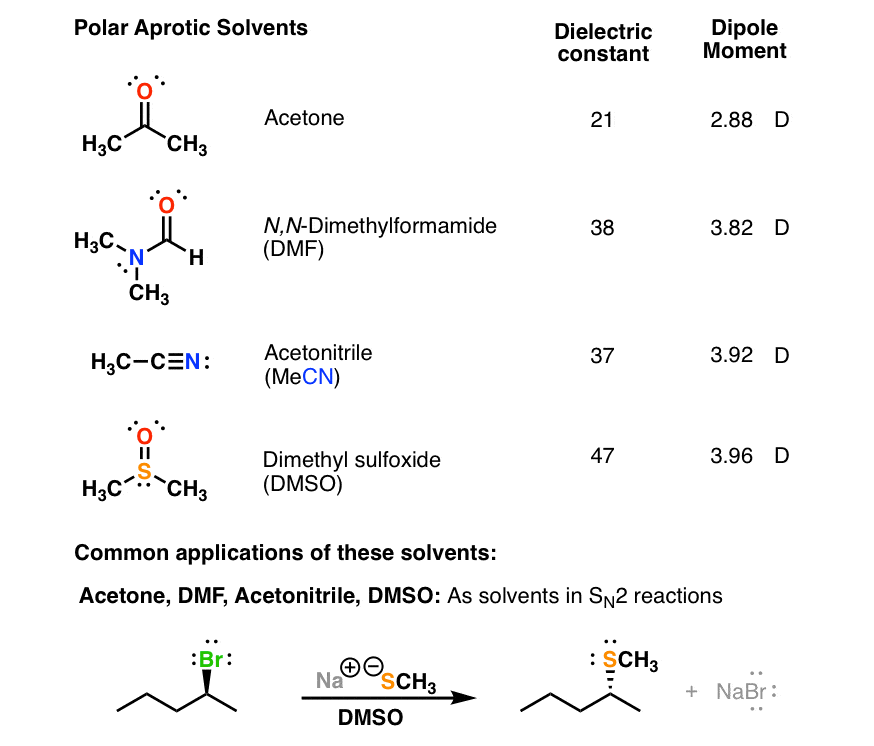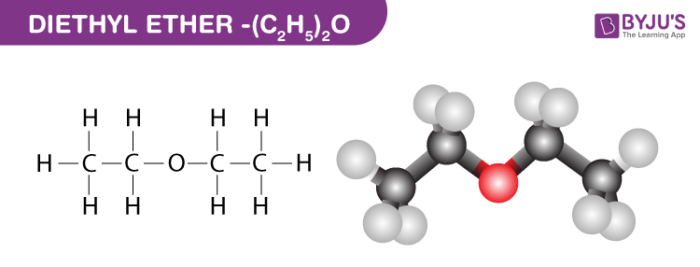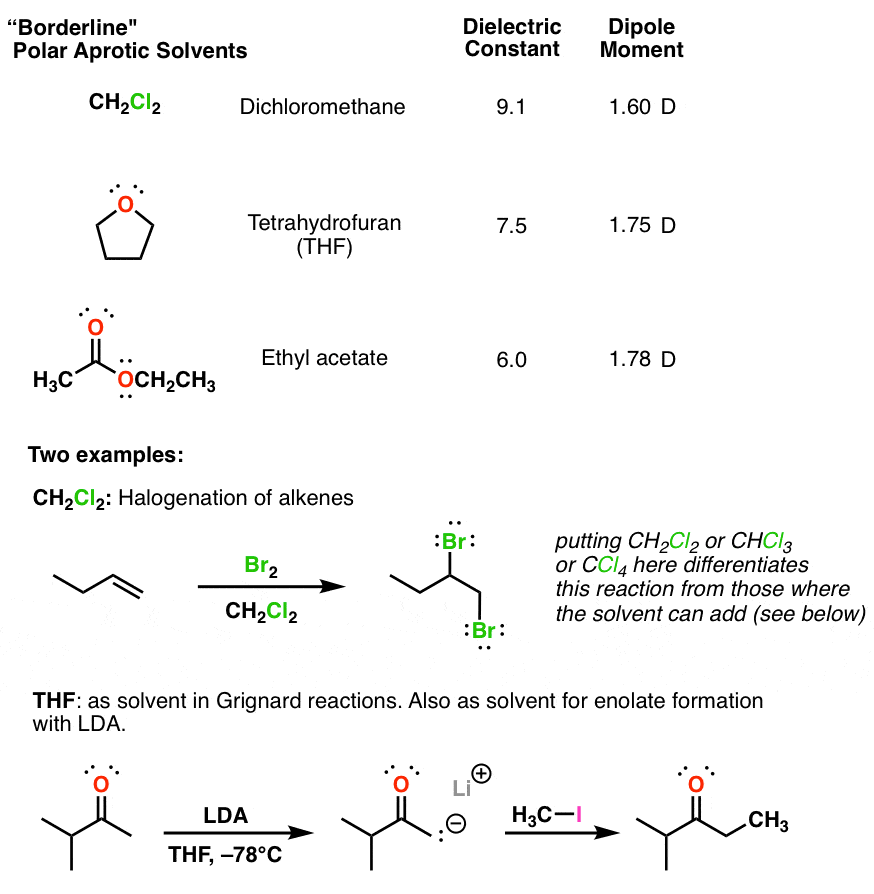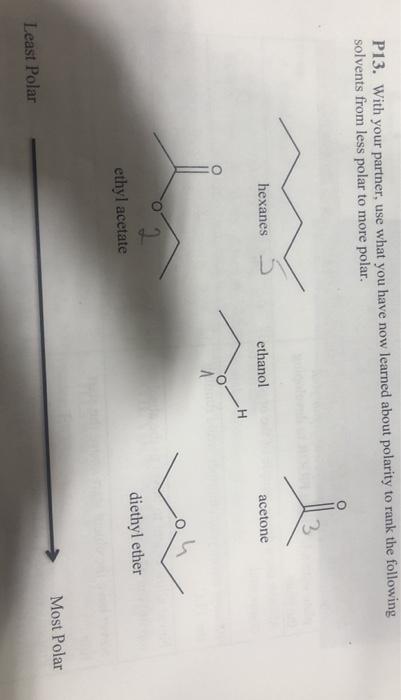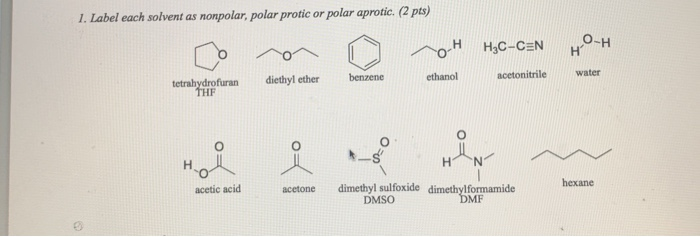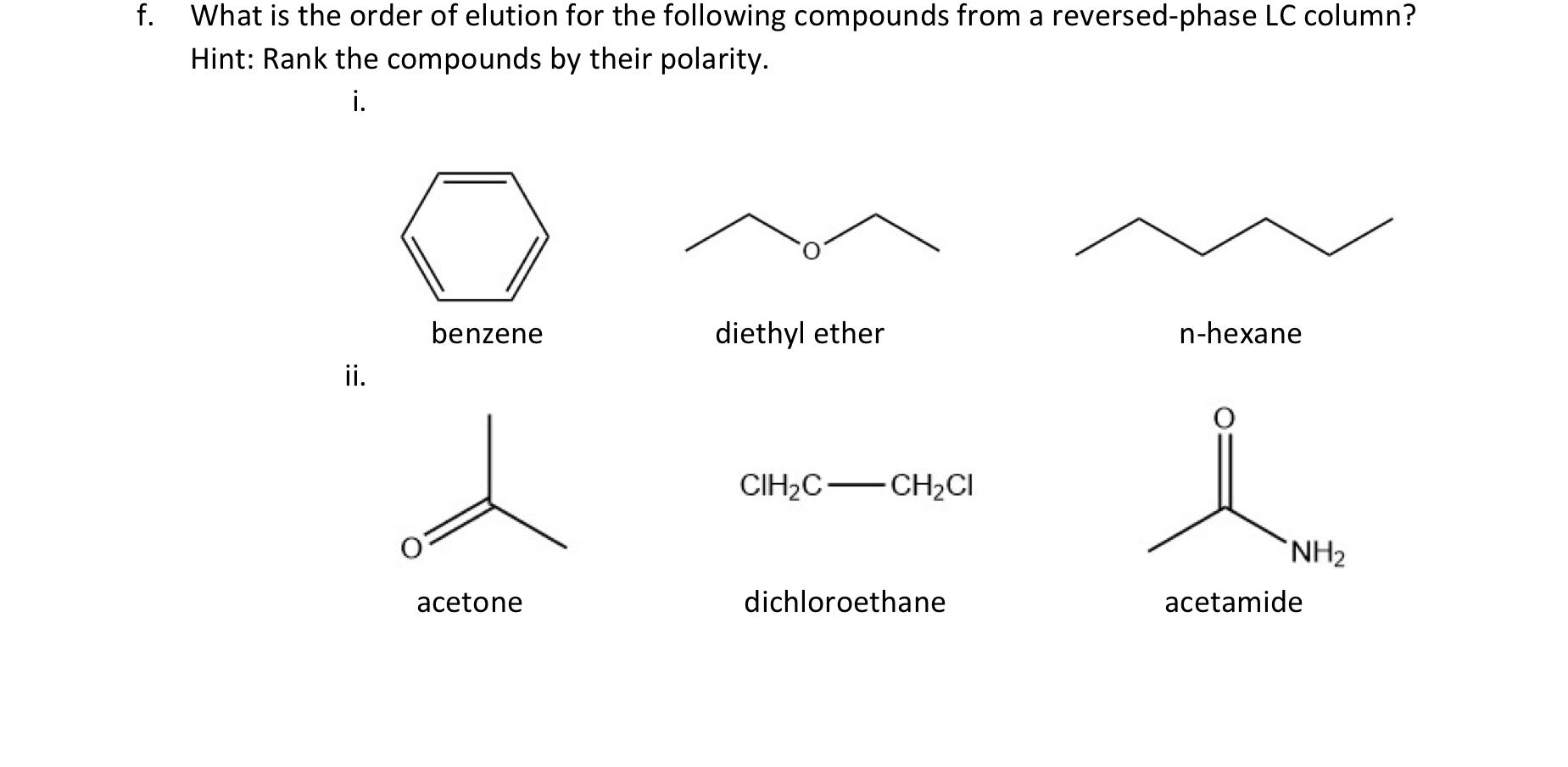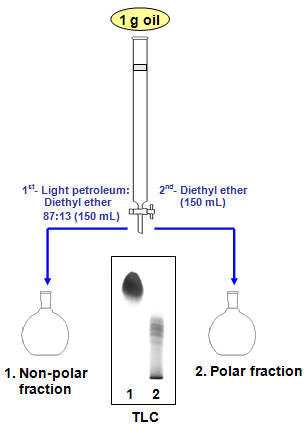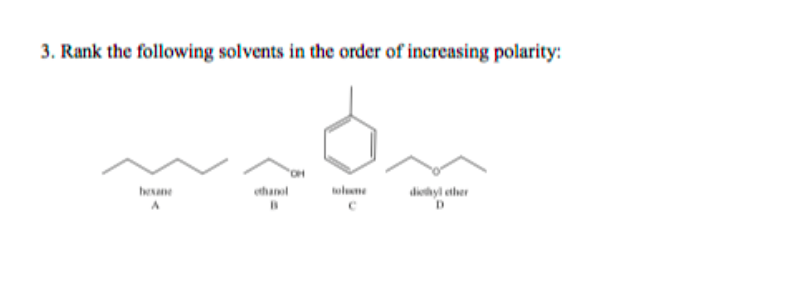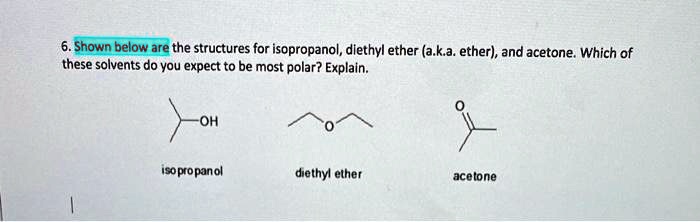
SOLVED: Shown below are the structures for isopropanol, diethyl ether (a.k.a. ether), ad acetone: Which of these solvents do vou expect to be most polar? Explaln; OH Isopropanol diethyl ether acetone
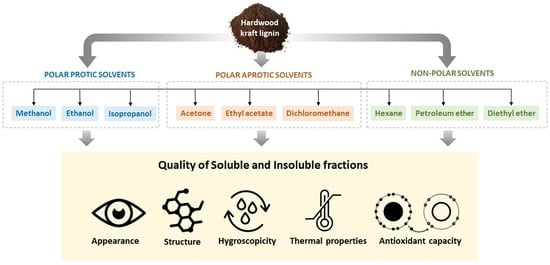
Polymers | Free Full-Text | One-Step Lignin Refining Process: The Influence of the Solvent Nature on the Properties and Quality of Fractions

What characteristics do methanol, chloroform, diethyl ether, etc. possess that makes them good solvents? - Quora

Solved: Determine The Solvent Polarity Index For The Follo... | Chegg.com | Index, Solvent, Homework help