Draw the Lewis dot structure for CCl4. Determine the electron geometry and molecular shape of this molecule. Is this molecule polar or nonpolar? | Homework.Study.com

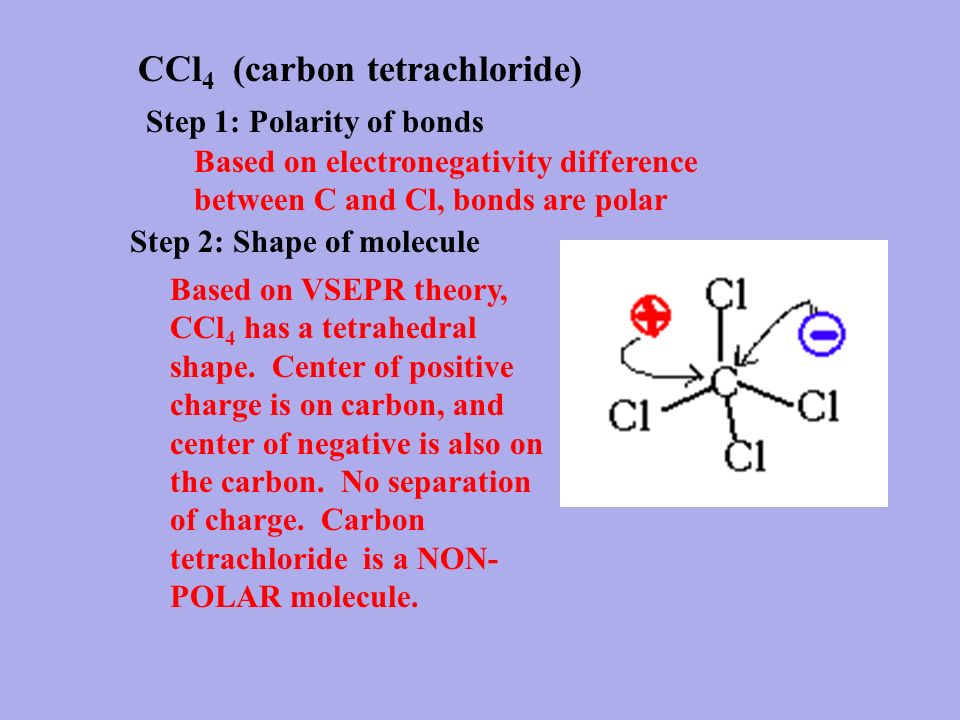
OneClass: Be sure to answer all parts The four bonds of carbon tetrachloride (CCI4) are polar, but th...

Draw the structure of carbon tetrachloride according to Lewis theory. What would be its associated molecular geometry? (a) tetrahedral (b) linear (c) trigonal pyramidal (d) bent | Homework.Study.com

Ozone in Polar (Acetonitrile) and Nonpolar (Carbon Tetrachloride) Organic Liquids: Optical Absorption, Solubility, and Stability - Ershov - 2022 - ChemistrySelect - Wiley Online Library

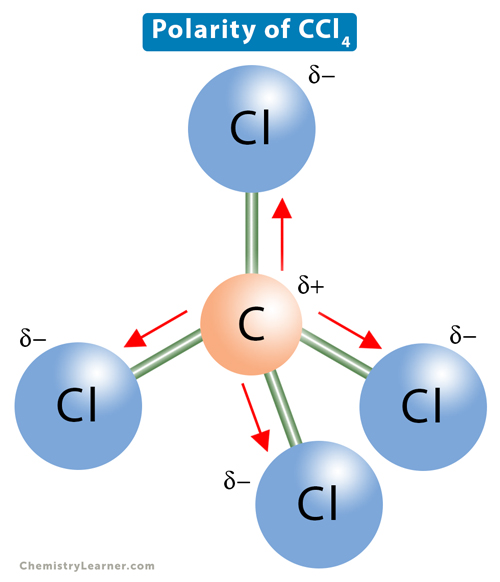
SOLVED: Explain how a molecule that contains polar bonds can be nonpolar. In your answer, use carbon tetrachloride, CCl4, as an example.





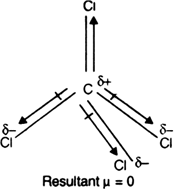
![Is \\[CC{l_4}\\] polar or nonpolar? Is \\[CC{l_4}\\] polar or nonpolar?](https://www.vedantu.com/question-sets/dbf3f5ee-35e7-43b7-b8a3-cfe63dfd520e2097468885289678636.png)