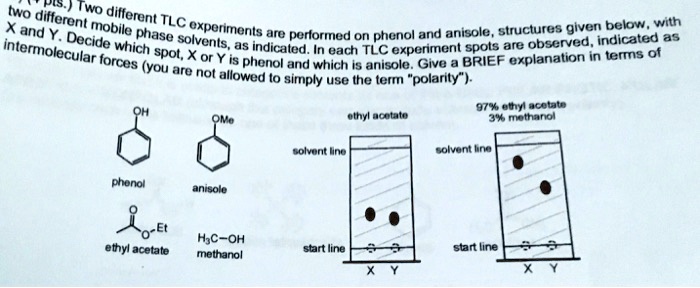
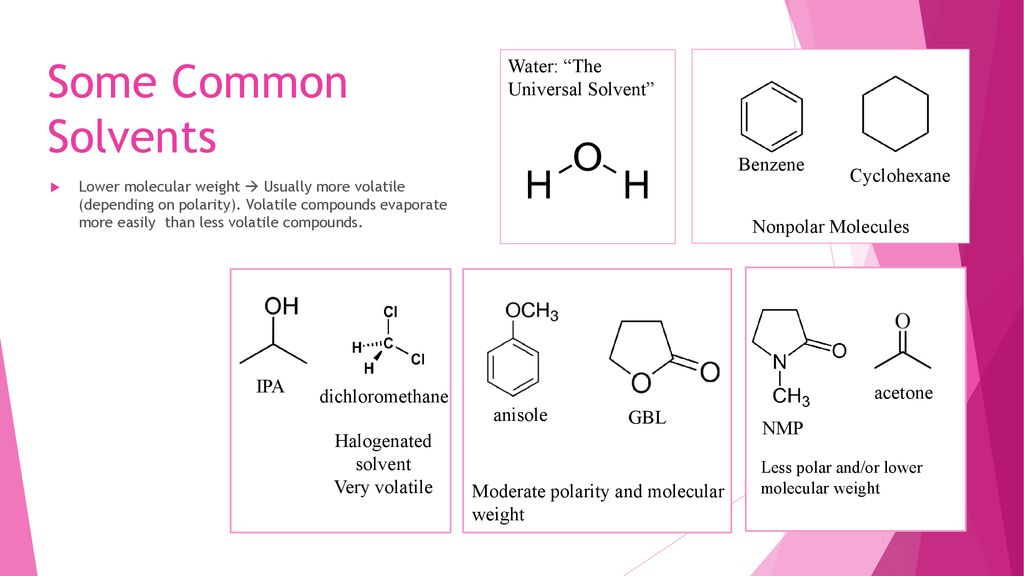
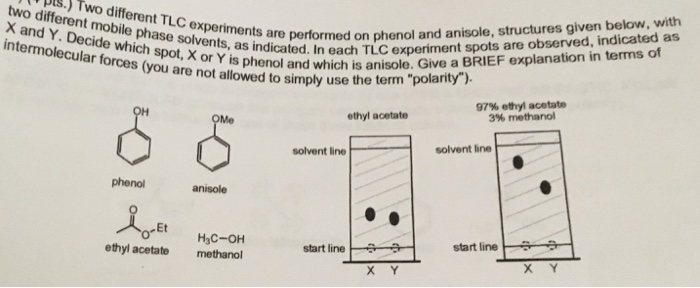
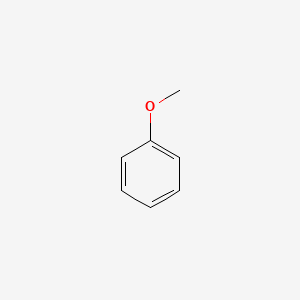
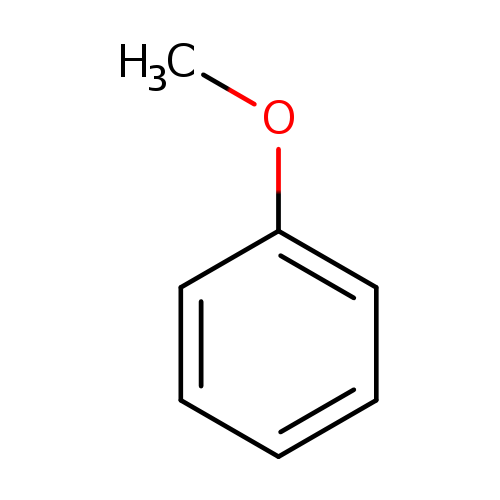
Anisole Hydrodeoxygenation over Nickel-Based Catalysts: Influences of Solvent and Support Properties | Energy & Fuels

WO2007041134A2 - Computer method and system for predicting physical properties using a conceptual segment model - Google Patents

Secondary organic aerosol formation from atmospheric reactions of anisole and associated health effects - ScienceDirect

Effects of surfactant on the molecules of different polarity of solubilization: Based on the study of micellar microscopic morphology mechanism - ScienceDirect
The mechanism of the Birch reduction. Part 2: a transition state model. - Henry Rzepa's Blog Henry Rzepa's Blog

Exciton-to-trion conversion as a control mechanism for valley polarization in room-temperature monolayer WS2 | Scientific Reports
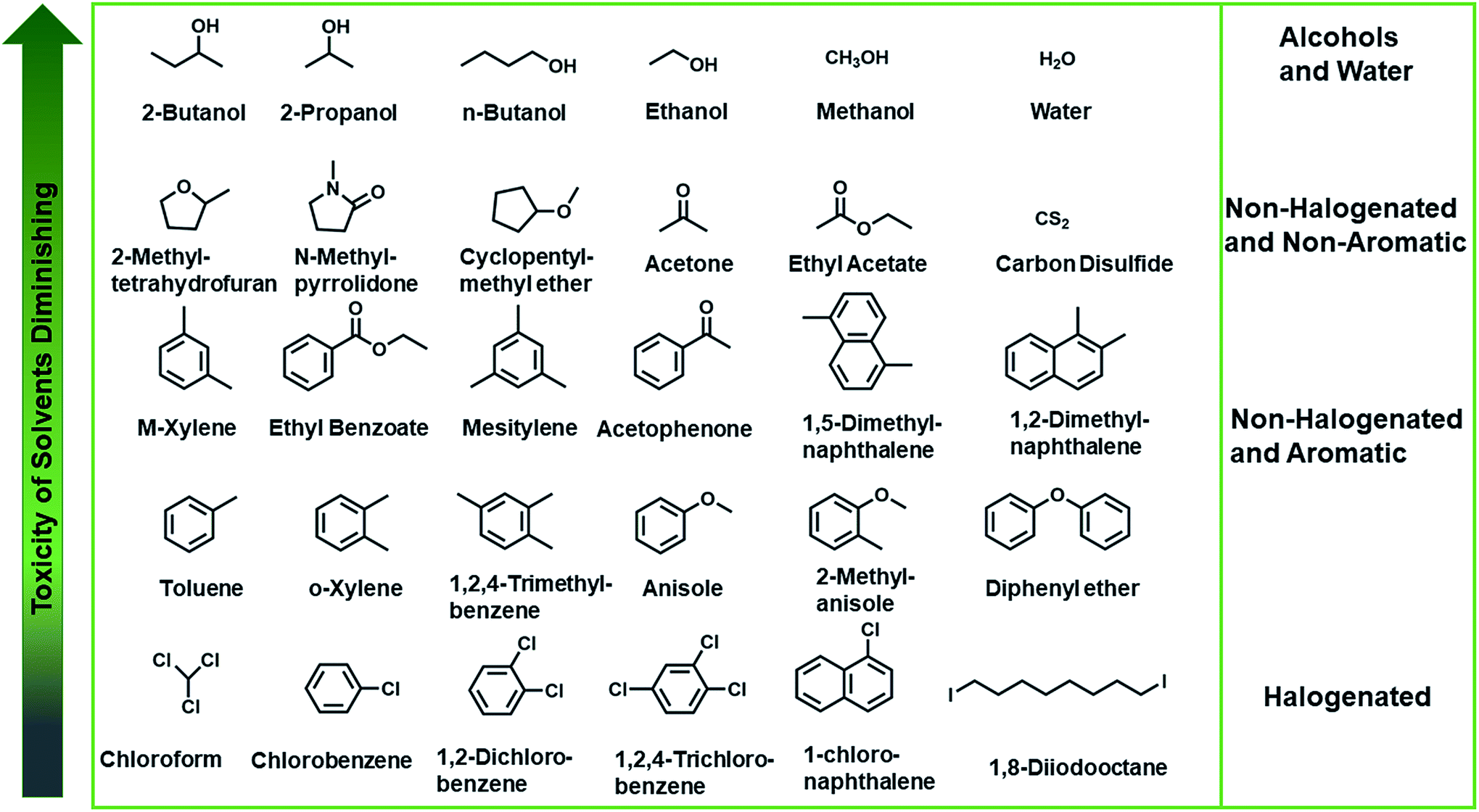
Green-solvent-processable strategies for achieving large-scale manufacture of organic photovoltaics - Journal of Materials Chemistry A (RSC Publishing) DOI:10.1039/C9TA09277C

Anisole Hydrodeoxygenation over Nickel-Based Catalysts: Influences of Solvent and Support Properties | Energy & Fuels
How was a mixture of organic molecules (phenol, benzoic acid, p-toluidine and anisole) separated and isolating process? - Quora